Phase 3 Asset
Derived from placental tissues, MOTYS™ is a biologic therapeutic classified as a Regenerative Medicine Advanced Therapy (RMAT). Effects from a single intra-articular injection of MOTYS™ last 9-12 months—no other treatment is this potent.
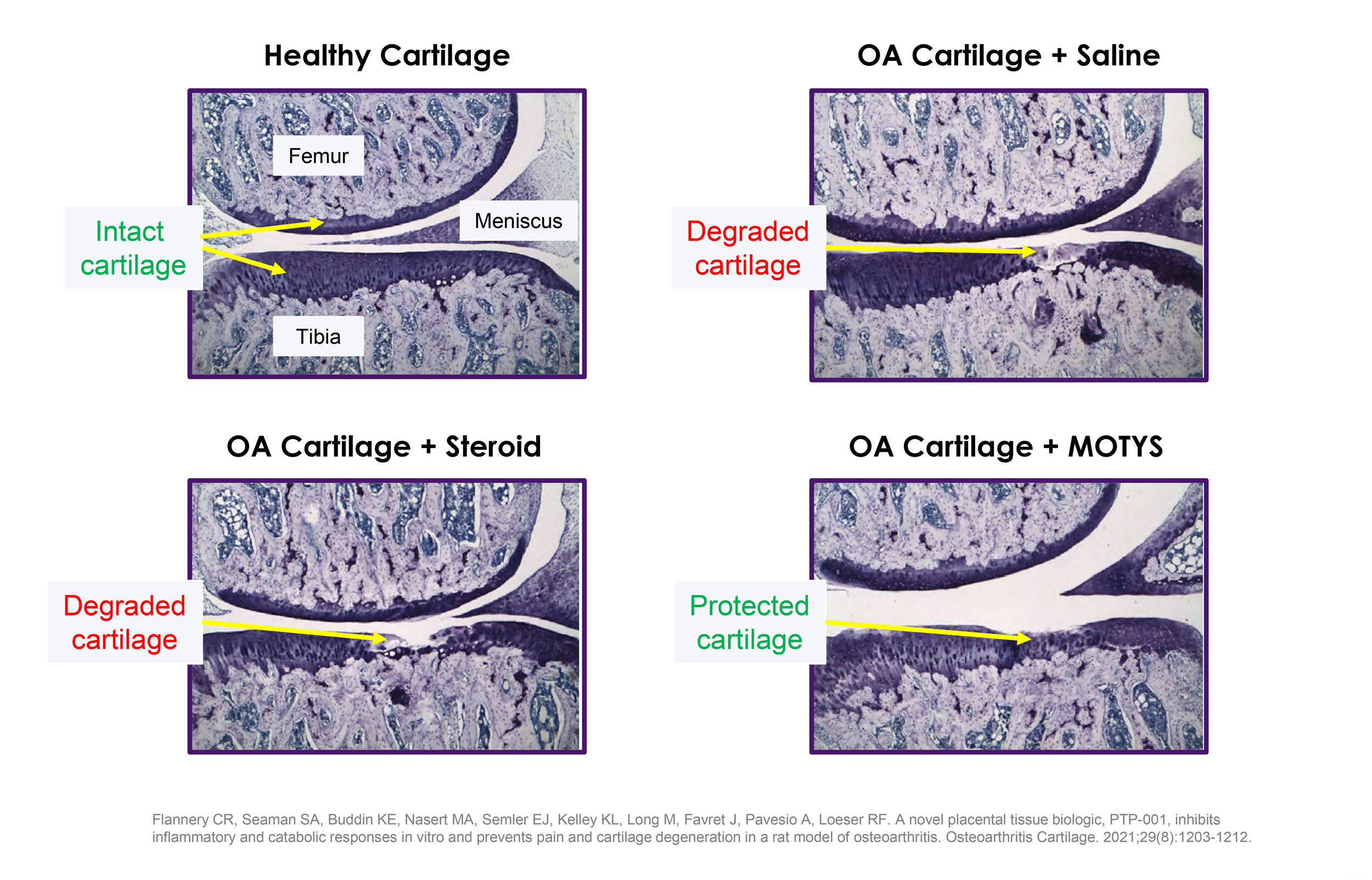
MOTYS™ holds great potential as a biologic therapy, because it displays a multifactorial mechanism of action (MOA). This MOA promotes a beneficial combination of anti-inflammatory, anti-catabolic and pro-anabolic effects, demonstrated through in vitro and in vivo data in a range of applications.
Pro-anabolic
New tissue formation through cell recruitment, differentiation, and/or proliferation
Anti-catabolic
Mitigation of cartilage degeneration
Anti-inflammatory
Reduction of pain and swelling